
The European Medicines Agency (EMA) is delivering a Product Management Service (PMS) and a Substance Management Service (SMS) to support regulatory activities in the European Union (EU) | Pharmya

European Medicines Agency approves pembrolizumab plus axitinib for first-line treatment of advanced kidney cancer - Action Kidney Cancer

European Medicines Agency (EMA) launches pilot project on drug repurposing – EJP RD – European Joint Programme on Rare Diseases

European Medicines Agency (EMA) | CDE Almería - Centro de Documentación Europea - Universidad de Almería

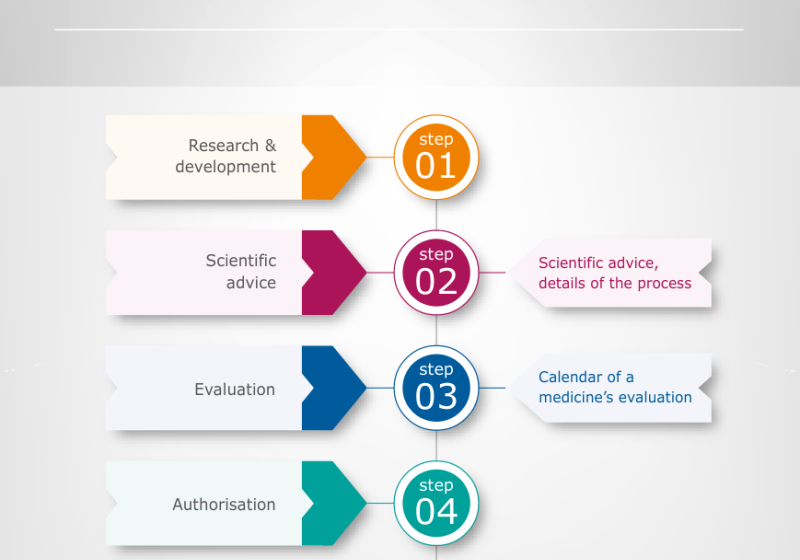





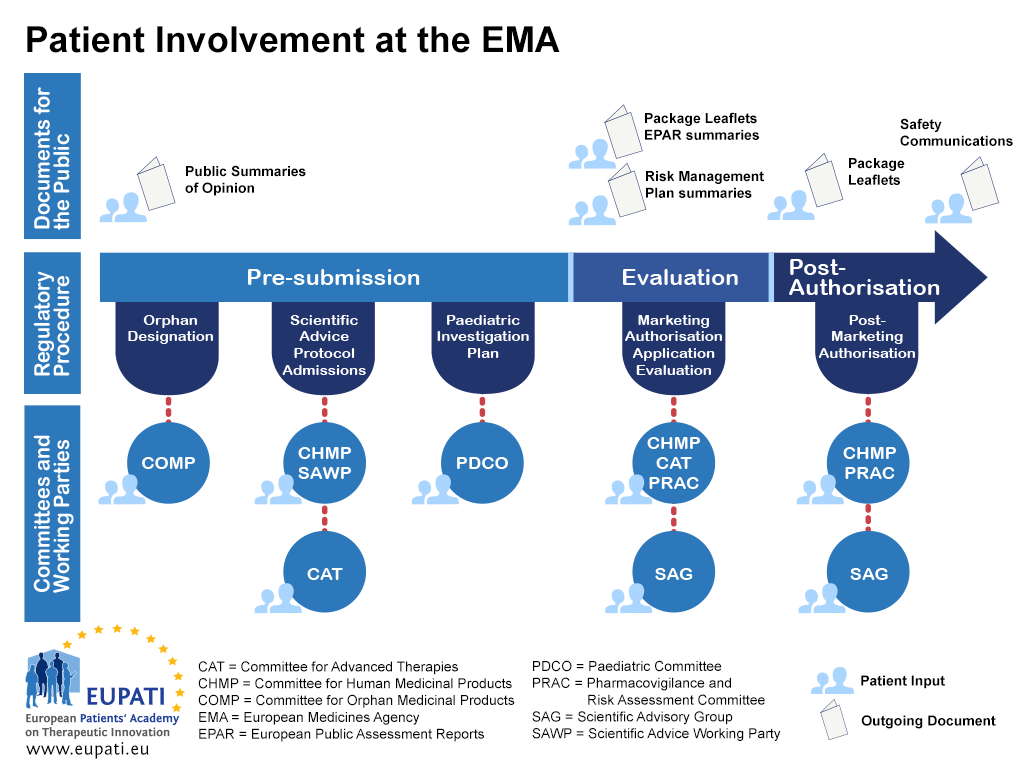




.jpg)

/cloudfront-us-east-2.images.arcpublishing.com/reuters/BIWQBPADDROF5LP7NMSSMUCRLY.jpg)






