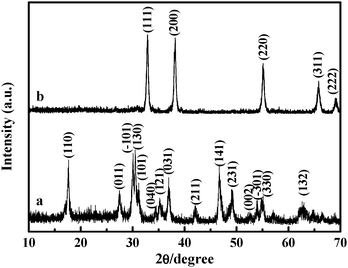
Structural parameters of Cd(OH) 2 calculated by XRD data prepared by... | Download Scientific Diagram

The molar solubility of Cd(OH)(2) is 1.84xx10^(-5)M in water. The expected solubility of Cd(OH)(2) in a buffer solution of pH = 12 is
One-step fabrication of Cd(OH)2 nanorings via a solution phase synthesis - Chemical Communications (RSC Publishing) DOI:10.1039/C0CC00665C

Solubility of `Cd(OH)_(2)` in pure water is `1.84xx10^(-5)\"mole\"//L` Calculate its solubility in - YouTube

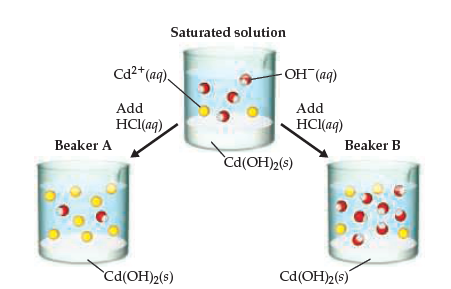
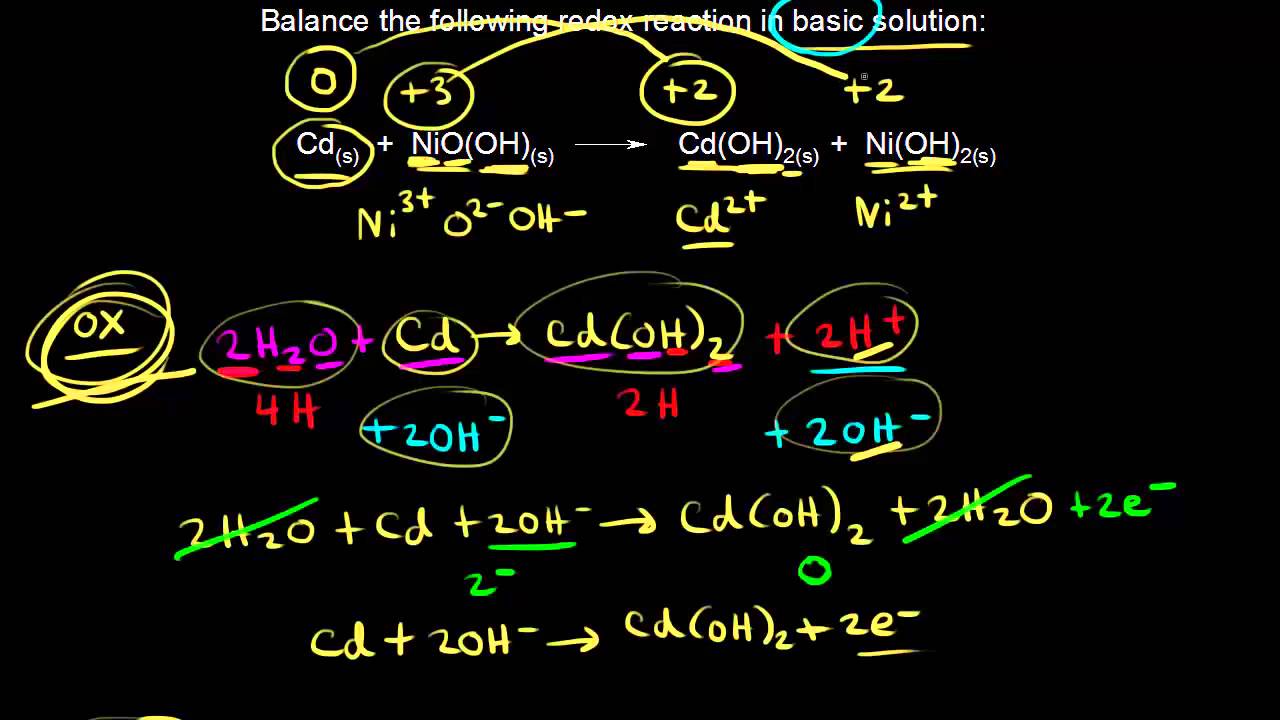
SOLVED:Cd(OH)2 is an insoluble compound. It dissolves in excess NaOH in solution. Write a balanced ionic equation for this reaction. What type of reaction is this?

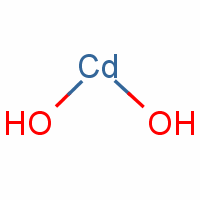
Buy high pure Cadmium hydroxide Cd(OH)2 Cd(OH) 99.9% chemical compound material 99.9% schpm Industrial Grade from Sichuan HPM - ECHEMI